IVC Filter Lawsuit
Are IVC Filters Deadly for Some Patients?
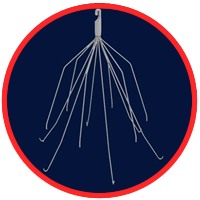
A recent study outlined that the use of inferior vena cava filters (IVC) in some pattens could more than double the mortality risk compared with similar patents who do not receive the device. questions about whether IVC filters are safe for use. In one IVC filter lawsuits the manufacture was ordered to pay $3.6 million to a woman who alleged she suffered serious complications because of an IVC filter.
What is an IVC filter?
IVC filters were designed to prevent blood clots from traveling to the patients lungs. These filters are implanted in the inferior vena cava (a vein moving blood from the lower body to the heart) during a surgery. The spider-like device with protruding legs is designed to catch blood blots from making their way to the heart or lungs.
IVC filters implanted into Patients?
IVC filters are implanted into patients who show risk of deep vein thrombosis. These short term use IVC filters are meant to be implanted temporarily (and then retrieved ) in a patient who have a short-term risk such as a patient suffering from a traumatic injury (car accident, etc.). Permeant IVC filters are designed for long-term use and designed for leaving the IVC filter implanted into the body for a longer period of time. If the an IVC filter is left in the body for longer than its intended use it may increases the risk that of IVC filter may fail.
Complications associated of IVC filters?
There are a variety of complications linked to the IVC filter:
- The filter can migrate
- The filter can cause thrombosis
- The folder can fracture
- The filter can cause ripping or damage to the vein
These complications can cause injuries or permanent damage.
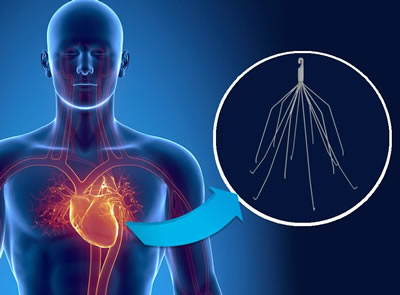
Filter Migration:
The IVC filter moves form the location where it was implanted which can cause it to block blood flow, damage the inferior vena cava, or other it can cause other significant harm and damage if it migrates. If the filter migrates from its original location it can be very difficult for a doctor to remove, it can increases the time the filter remains in the body and it can dramatically increase the risk of complications. If the filter comes loose in the body and migrates to the patients heat or lungs it can cause life-threatening complications.
Filter Fracture:
The IVC filter can also fracture where part of the filter bends, fractures or breaks off in the body. The leg or strut of the IVC filter is what typically breaks off and may perforate the patient’s IVC walls or migrate to a different position, also increasing the risk of harm and damage.
Filter Perforation:
The IVC filter due to its struts or legs can rip into or can puncture a blood vessel or worst an internal organ.
If an IVC Filter failure symptoms:
- Chest pain
- Neck pain
- Abdominal pain
- Back pain
- Leg pain
- Confusion or Lightheadedness
- Shortness of breath
- Rapid heart rate
- Nausea or vomiting
If the IVC filter migrates and cannot be removed, it can cause serious health problems:
- Hole in the heart
- Arrhythmia
- Stroke
- Internal bleeding

The U.S. Food and Drug Administration (FDA) in 2010 issued a safety warning and communication after receiving reports of adverse events linked to the filters in five years.The FDA’s warning included recommending that filters be removed as soon as the risk of pulmonary embolism subsides but in some cases, the filter it is claimed that the filter was left in the patient longer than was necessary.
In 2014, the FDA updated its safety communication specifically, the FDA updated information related to published research and post-market surveillance studies for the medical devices which include further information about the risk of serious complications linked to IVC filters.
The FDA warned, among other items that IVC Filters are linked migration, fracture, embolization (where the device or part of the device moves to the heart or lungs), perforation, and difficulty removing the device. The FDA noted that once the short-term risk of pulmonary embolism has passed, IVC filters should ideally be removed between 30 – 60 days after they were implanted.
IVC filter lawsuits?
IVC filter lawsuits have been filed across the United States alleging that manufacturers knew or should have known about the risks associated with this medical device and failed to warn patients or the medical community about the risks. They further allege negligence on the part of the manufacturer or marketer and design and manufacturing defects.
Did you or a loved one have complications with an IVC filter?
Our partners law firms are looking into claims that the manufactures of IVC Filters knew about defects in the products that caused damage to patients. If you or a loved one received an IVC Filter product made by C.R. Bard, Cook Medical, Boston Scientific or another company and had complications you may be eligible to file a lawsuit against the manufacture which could help you recover compensation for:
- Pain and suffering
- Punitive damages
- Past and future lost wages
- Medical bills
- Disability
- Other damages
100% Free Information, you will not be asked to pay anything.
Take action today, free case evaluation.
First Name
Last Name

Email Address

Phone Number

Address

City

State

Zip Code

Comments

